 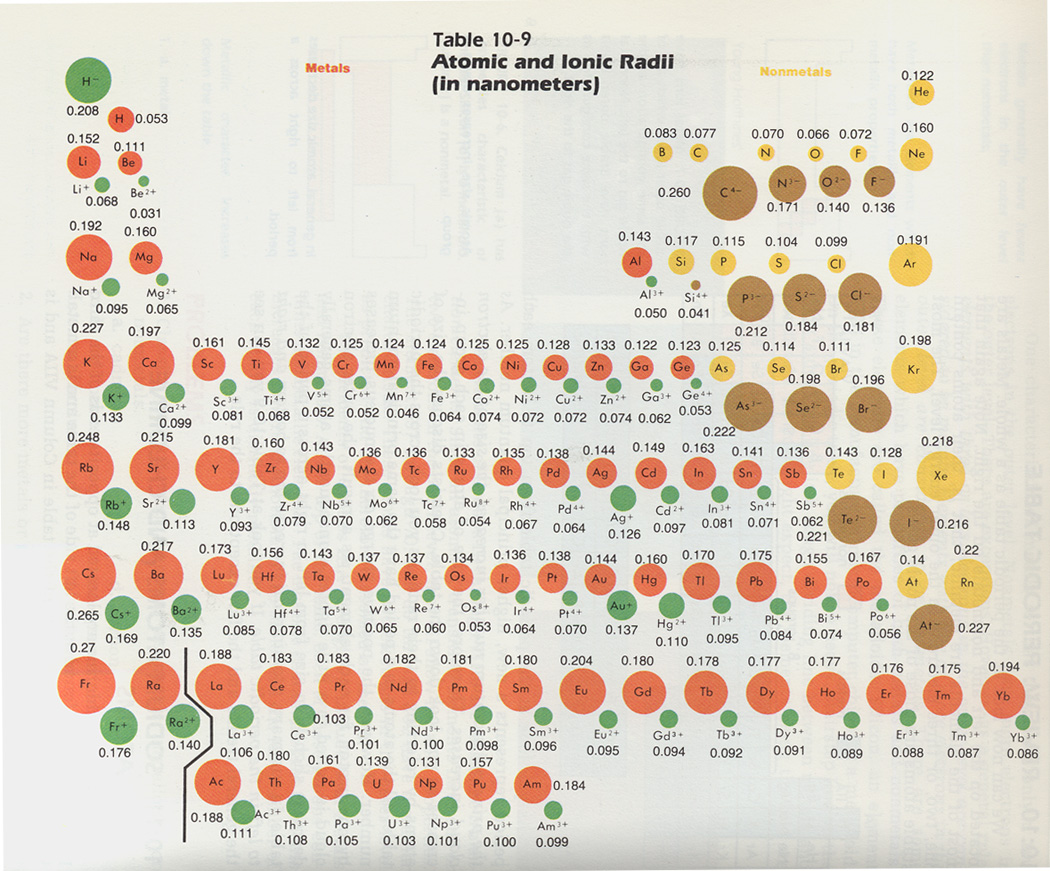
Yeah, He is even smaller than hydrogen, H, which is 53 pm. Since 0.85 Å is the only option that is less than 1.06 Å, the correct option is 'd'. The smallest atom on the periodic table is helium, He, and has a radius of 31 pm. In lanthanoids, the atomic and ionic radii decrease with increase in atomic number. Lanthanum is the first element and Lutetium is the last one. Lanthanum, La and Lutetium, Lu belong to lanthanoids (4f block elements of inner transition elements). Which one of the following given values will be closest to the radius of Lu 3+ (atomic number : Lu =71) ? 6) The radius of La 3+ (atomic number : La=57) is 1.06 Å. The correct order of ionic radii is Na + > Mg 2+ > Al 3+ > Si 4+ as given in option "4". The correct option is "d" 5) Which of the following is correct order of ionic radii?įor isoelectronic ions, the radius decreases with increase in positive charge. atomic radius, half the distance between the nuclei of identical neighbouring atoms in the solid form of an element. For different ions of same element, size decreases with increase in the positive charge. The radius of an atom may be taken as the distance between atomic nucleus and the outermost shell of electrons of the atom. K + and Cl - are isoelectronic species but the number of protons are more in K +. Since the proton number is same in isotopes, the nuclear attraction is also same. These ions belong to two different isotopes of same element. The size decrease with increase in the positive charge these species. The atomic size increases from top to bottom in a group. Size decreases with increase in proton number The ionic radii increases with decrease in the effective nuclear charge. Generalization: In iso-electronic species, the atomic size decreases with increase in the atomic number. 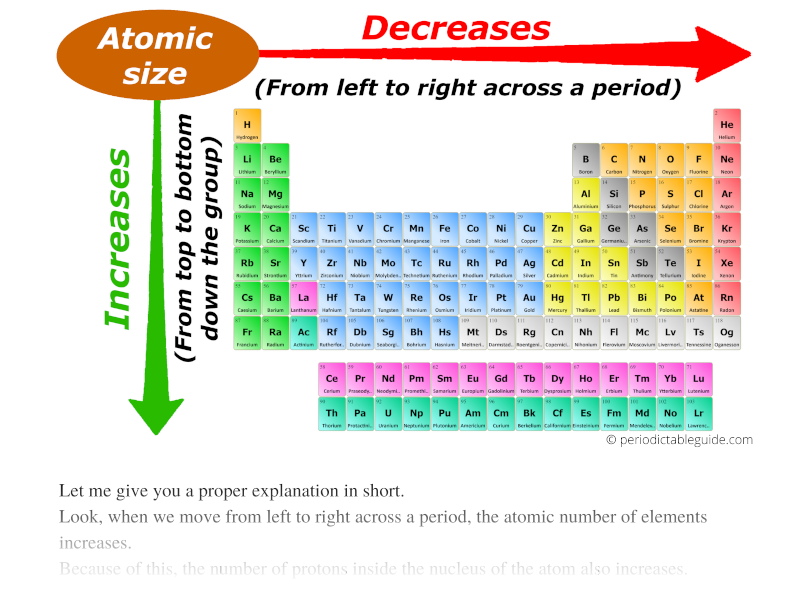
Hence the attraction is minimum and the ion is largest. However, in N 3- ion, there is only 0.7 proton for each electron. Hence the attraction is maximum and the ion is smallest among the given species. Greater this value, greater is the attraction and smaller is the size.įor example, in Na + ion, there is 1.1 proton for each electron. However, going from left to right will actually decrease. of protons (Z) to number of electrons correlates effective nuclear attraction. Most people clearly understand that atomic radius will increase as you go down the periodic table. Related questions 2) Which of the following is the smallest in size? Note: It is not possible to get covalent and metallic radii for noble gases since they do not form bonds.Ībove fact is reflected in option "a". Hence neon's atomic radius must be much more than that of fluorine. The reported radii of noble gas elements are "van der Waals radii", which are 40% more than the actual atomic radii. PERIODIC TRENDS IN RADIUS OF ATOM AND IONĪdiChemistry IIT JEE Atomic radii of fluorine and neon in Angstrom units are respectively given by: Questions periodic trends | atomic radius | IIT JEE | NEET 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
